Kidney Disease Quality Measure Development, Maintenance, and Support
Principal Investigator: Joseph Messana

Under this contract, UM-KECC provides CMS with the support necessary to revise and enhance quality measurement within the CKD and ESRD setting. This support includes work to develop, maintain and update the technical specifications for current and future ESRD measures. Once measures are in use, CMS requires that they periodically be reevaluated. In addition, UM-KECC will maintain and calculate the quality measures publicly reported on the Dialysis Facility Compare (DFC) website, a venue which allows patients, families and others to compare dialysis facilities on a number of characteristics and quality measures.
This Task Order is one of the initial task orders competed under the Measure & Instrument Development and Support (MIDS) IDIQ Umbrella contract for 2018-2028. MIDS program promotes the quality of healthcare by emphasizing major strategies for evaluating and improving care. Under multiple task orders, the MIDS contractors will assist the Centers for Medicare and Medicaid Services (CMS) in developing
ESRD Data Utilization: Dialysis Facility Reports (Oversights/DFR)
Principal Investigator: Joseph Messana
 Under this contract, UM-KECC creates detailed data reports about End Stage Renal Disease
(ESRD) dialysis facilities, Transplant Programs, and Organ Procurement Organizations
(OPOs) in formats that are useful to the Centers for Medicare & Medicaid Services
(CMS), States, health care providers and the general public. These reports will include
summaries of patient characteristics, facility characteristics, patient outcomes,
and facility practice patterns. These reports will include comparative State, Network,
Regional, and National statistics as appropriate. These reports will be the basis
for overseeing the quality and safety of the ESRD, Transplant and OPO Programs within
CMS.
Under this contract, UM-KECC creates detailed data reports about End Stage Renal Disease
(ESRD) dialysis facilities, Transplant Programs, and Organ Procurement Organizations
(OPOs) in formats that are useful to the Centers for Medicare & Medicaid Services
(CMS), States, health care providers and the general public. These reports will include
summaries of patient characteristics, facility characteristics, patient outcomes,
and facility practice patterns. These reports will include comparative State, Network,
Regional, and National statistics as appropriate. These reports will be the basis
for overseeing the quality and safety of the ESRD, Transplant and OPO Programs within
CMS.
The Evaluation of the End-Stage Renal Disease Treatment
Choices Model and Kidney Care Choices Model
Co-Principal Investigators: Claudia Dahlerus and Richard A. Hirth
 The Centers for Medicare and Medicaid Services (CMS) Innovation Center is testing
two new care delivery models aimed at improving the quality of care for people living
with kidney disease. This project is an independent evaluation of these models. In
collaboration with The Lewin Group (Sponsor) the University of Michigan (subcontractor)
is serving on the independent evaluation team for these two new models. The End Stage
Renal Disease (ESRD) Treatment Choices (ETC) Model includes incentives for kidney
doctors and dialysis clinics to increase use of home dialysis and increase successful
kidney transplants. The other new approach is the Kidney Care Choices (KCC) Model
which includes incentives for kidney doctors to slow down chronic kidney disease (CKD),
increase home dialysis, and shared incentives for kidney doctors and transplant providers
to increase kidney transplantation. The aims of the Evaluation are to assess how well
model participants (nephrology practices along with dialysis facilities, transplant
providers) were able to increase patient uptake of home dialysis; increase access
to kidney transplant, and for the KCC Model, slow CKD progression to ESRD. UM-KECC
will analyze metrics assessing CKD progression and access to transplant, as well as
interviewing providers participating in the KCC Model, kidney disease patients, patient
care partners, and potential living donors on how they think the Models are doing
in achieving the above stated goals.
The Centers for Medicare and Medicaid Services (CMS) Innovation Center is testing
two new care delivery models aimed at improving the quality of care for people living
with kidney disease. This project is an independent evaluation of these models. In
collaboration with The Lewin Group (Sponsor) the University of Michigan (subcontractor)
is serving on the independent evaluation team for these two new models. The End Stage
Renal Disease (ESRD) Treatment Choices (ETC) Model includes incentives for kidney
doctors and dialysis clinics to increase use of home dialysis and increase successful
kidney transplants. The other new approach is the Kidney Care Choices (KCC) Model
which includes incentives for kidney doctors to slow down chronic kidney disease (CKD),
increase home dialysis, and shared incentives for kidney doctors and transplant providers
to increase kidney transplantation. The aims of the Evaluation are to assess how well
model participants (nephrology practices along with dialysis facilities, transplant
providers) were able to increase patient uptake of home dialysis; increase access
to kidney transplant, and for the KCC Model, slow CKD progression to ESRD. UM-KECC
will analyze metrics assessing CKD progression and access to transplant, as well as
interviewing providers participating in the KCC Model, kidney disease patients, patient
care partners, and potential living donors on how they think the Models are doing
in achieving the above stated goals.
End Stage Renal Disease Quality Incentive Program (ESRD QIP) Support
Principal Investigator: Yi Li
 UM-KECC serves as a subcontractor to Arbor Research to provide support for the CMS
ESRD Quality Incentive Program (QIP) that was first implemented for Payment Year (PY)
2012 to guard against any potential unintended consequences and a decline in the quality
of care under the new ESRD bundled payment system, CMS’s first national value-based
purchasing program.
UM-KECC serves as a subcontractor to Arbor Research to provide support for the CMS
ESRD Quality Incentive Program (QIP) that was first implemented for Payment Year (PY)
2012 to guard against any potential unintended consequences and a decline in the quality
of care under the new ESRD bundled payment system, CMS’s first national value-based
purchasing program.
CDC Chronic Kidney Disease (CKD) Surveillance System
Principal Investigator: Rajiv Saran
 UM Nephrology in collaboration with the Centers for Disease Control & Prevention and
University of California, San Francisco, has successfully established a Chronic Kidney
Disease Surveillance System for the entire United States. It systematically tracks;
reports trends in burden; awareness and impact on population health in the following
six major topics: (1) the burden of disease, (2) burden of risk factors, (3) disease
awareness, (4) quality and processes of care, (5) health consequences associated with
CKD and (6) health system capacity available to deal with CKD.
UM Nephrology in collaboration with the Centers for Disease Control & Prevention and
University of California, San Francisco, has successfully established a Chronic Kidney
Disease Surveillance System for the entire United States. It systematically tracks;
reports trends in burden; awareness and impact on population health in the following
six major topics: (1) the burden of disease, (2) burden of risk factors, (3) disease
awareness, (4) quality and processes of care, (5) health consequences associated with
CKD and (6) health system capacity available to deal with CKD.
Optimization and Simulation of Kidney Paired Donation Programs
Principal Investigator: Jack D. Kalbfleisch
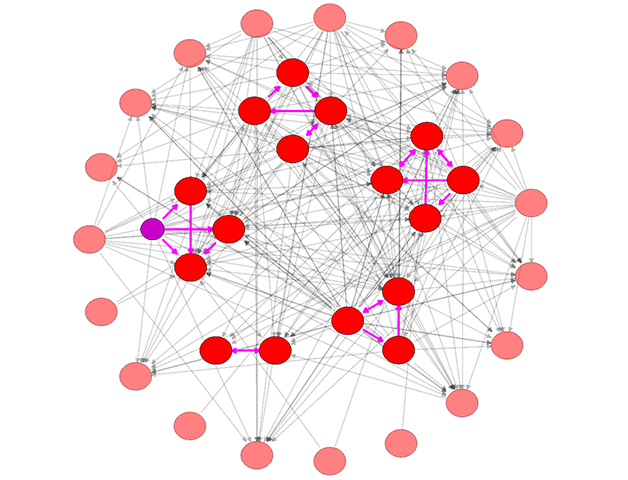
The above is an illustration of a KPD network, with pairs represented by red nodes and altruistic donors represented by purple nodes. Edges connecting nodes represent donor-candidate matches based on a virtual (computer based) cross match. A selection with five subsets is highlighted and the possible transplants are indicated by purple edges. These subsets are locally relevant subsets of size four or less and are chosen to present fall back opportunities should some transplants not be viable. We proceed by evaluating all transplants for viability indicated by the purple arrows, then choosing the best possible disjoint set of cycles and chains for transplant. (See Bray et al., 2015)
Kidney paired donation (KPD) provides an approach to overcome the barriers faced by many patients with kidney failure who present with willing, but immunologically or blood type incompatible living donors. KPD programs use a computerized algorithm to match one incompatible donor/recipient pair to another pair with a complementary incompatibility, such that the donor of the first pair gives to the recipient of the second, and vice versa. More complex exchanges of organs involving three or more pairs are also considered as are altruistic or non-directed donors (NDD) who donate a kidney voluntarily and thereby have the potential to create a chain of kidney transplants. Such donors and chains have become increasingly important in KPD programs. Checking the viability of all potential transplants in a pool is not logistically possible, and so a fundamental problem in a KPD program is selecting an optimal subset of matches to consider among the many possibilities that exist. We are developing methods of selecting potential matches that take account of the uncertainty in the process; namely that potential transplants that are identified on a computer algorithm often fail when an attempt is made to put them into practice. Our approaches select subsets of patients with many options or fallbacks that could be implemented depending upon which potential transplants are found to be viable, and have been shown to have the potential to greatly increase the number and/or utility of transplants performed. We are also developing user friendly and efficient software to implement these approaches. In this work, we utilize data from the University of Michigan Paired Donation Program and the Alliance for Paired Donation.
Kidney Graft Survival Calculator:
Our calculator estimates the probability of graft survival given the characteristics
of the candidate and potential donor, based on a Cox proportional hazards model fitted
to SRTR data. This calculator has particular potential as a guide to candidates with
a compatible living donor who may be able to improve their predicted graft or patient
survival through participation in a KPD program.
Software:
KPD GUI: Software and Graphical Interface (download)
Our software platform can be used to manage and visualize exchanges suggested by optimization criteria in KPD, offering several advantages over other available software:
Interactive visual display of the state of the KPD
Implementation of optimization methods from previous literature, accounting for probabilities
of failure, as well as fallback options (uncertainties and contingencies)
Optimization extended to more general subsets of pairs and NDDs that facilitate fallback
options.
Posters:
Bray M, Wang W, Song PXK, Leichtman AB, Rees MA, Ashby VB, Eikstadt R, Kalbfleisch
JD. Incorporating Uncertainties and Contingencies in a Paired Donation Program. ASN
Kidney Week 2013. Atlanta, GA (November 7). American Society of Nephrology
Wang W, Bray M, Song PXK, Leichtman AB, Kalbfleisch JD. Multiple Decision Allocation Strategies in Kidney Paired Donation Program. ASN Kidney Week 2013. Atlanta, GA (November 7). American Society of Nephrology.
Bray M, Wang W, Song PXK, Kalbfleisch JD. Incorporating Transplant Candidates with Multiple Associated Incompatible Donors in Kidney Paired-Donation. ENAR Spring Meeting 2016. Austin, TX (March 6). Eastern North American Region of the International Biometrics Society.
Wang W, Bray M, Song PXK, Kalbfleisch JD. Locally Relevant Subgraph Enumeration in Transplant Patient Network. ENAR Spring Meeting 2016. Austin, TX (March 6). Eastern North American Region of the International Biometrics Society.
Related Projects
Optimization and Simulation of Kidney Paired Donation Programs
Kidney paired donation (KPD) provides an approach to overcome the barriers faced by many patients with kidney failure who present with willing, but immunologically or blood type incompatible living donors. KPD programs use a computerized algorithm to match one incompatible donor/recipient pair to another pair with a complementary incompatibility, such that the donor of the first pair gives to the recipient of the second, and vice versa. More complex exchanges of organs involving three or more pairs are also considered as are altruistic or non-directed donors (NDD) who donate a kidney voluntarily and thereby have the potential to create a chain of kidney transplants. Such donors and chains have become increasingly important in KPD programs. Checking the viability of all potential transplants in a pool is not logistically possible, and so a fundamental problem in a KPD program is selecting an optimal subset of matches to consider among the many possibilities that exist. We are developing methods of selecting potential matches that take account of the uncertainty in the process; namely that potential transplants that are identified on a computer algorithm often fail when an attempt is made to put them into practice. Our approaches select subsets of patients with many options or fallbacks that could be implemented depending upon which potential transplants are found to be viable, and have been shown to have the potential to greatly increase the number and/or utility of transplants performed. We are also developing user friendly and efficient software to implement these approaches. In this work, we utilize data from the University of Michigan Paired Donation Program and the Alliance for Paired Donation.
Enhancing the Cardiovascular Safety of Hemodialysis Care:
Patient-Centered Outcomes Research Institute
Principal Investigator: Rajiv Saran

Full Project Title: Enhancing the Cardiovascular Safety of Hemodialysis Care: A Cluster-Randomized, comparative Effectiveness Trial of Multimodal Provider Education and Patient Activation Interventions
Design: Cluster-Randomized controlled trial (CRCT) using Fresenius medical centers for the randomization, comparing two interventions to improve the CV safety of HD care by pursuing the following aims:
Aim 1 To translate two evidence-based interventions (multi-modal provider education
and patient activation using peer mentoring) from their prior application settings
into the context of outpatient hemodialysis (HD) care cardiovascular/hemodynamic safety.
Aim 2 Conduct a cluster-randomized controlled trial (CRCT) to compare the effects
of the above HD facility-level interventions on the primary outcome of dialysis session
safety over a period of 12 months.
Aim 3 Compare the effects of the two HD facility-level interventions on secondary
patient-centered clinical outcomes, including: patient symptoms, fluid adherence,
dialysis adherence, quality of life, hospitalizations and mortality over the same
timeframe.
Aim 4 Identify factors associated with successful implementation of the interventions,
and conduct analyses of mediators and moderators associated with primary and secondary
outcomes.
Reducing Kidney Disease Through Acute Kidney
Injury Risk Prediction and Prevention
Principal Investigator: Michael Heung

The study aims to a) develop a high fidelity AKI risk prediction model for hospitalized
Veterans; and b) develop an accompanying clinical decision support system (CDSS) to
provide
management recommendations for patients identified as being at higher risk for AKI.
A future goal would be to incorporate these tools into the VA electronic health record
(EHR) and to perform usability testing to establish their efficacy and impact.
Prediction Analytics to Guide Prevention of Kidney Disease Among U.S. Veterans
Principal Investigator: Rajiv Saran

The goal the study is to develop a data-driven approach to reduce the incidence and progression of kidney disease among Veterans in the U.S. The study propose is to carry out predictive analytics (using traditional statistical and machine learning tools) and geospatial analyses, to identify high risk individuals as well as those with identifiable kidney disease, and the geographic areas they live in, to better understand risk and progression factors for kidney disease. This work will set the stage for enabling targeted population health interventions to prevent kidney disease as well as slow progression of kidney disease among U.S. veterans.
Improve Statistical Methods for Profiling of Healthcare Providers
Principal Investigator: ZHI (KEVIN) HE
Healthcare provider (such as dialysis facilities and transplant centers) profiling is of nationwide importance. The statistical analysis along with further reviews may lead to changes in budgeting allocations and even suspension for providers with extremely poor outcomes. Existing profiling approaches typically assume that the risk adjustment is perfect, and the between-provider variation is entirely due to the quality of care, which, however, is often invalid. In addition, analyzing large-scale databases introduces computational difficulties, particularly when the event of interest is recurrent, and the numbers of sample size and the dimension of parameters are large. How to integrate streaming recurrent event data adds another level of difficulty. In view of these difficulties. Finally, the COVID-19 pandemic has dramatically changed how healthcare care is delivered. It is crucial that the profiling methods account for temporal and geospatial variation and accurately evaluate providers’ performance during the pandemic. The goal of this project is to develop a new class of risk adjustment models and statistical inference procedures to ensure an “apples-to-apples” comparison of healthcare providers.