Good Science Changes

By Amy Crawford
For a brief time, in very recent history, scientists believed they had discovered a biological marker in our chromosomes that could tell us how much longer we had to live.
 Telomeres are regions of repetitive nucleotides that cap the ends of chromosomes,
protecting DNA from degradation as cells divide and replicate. First identified in
the 1930s, their structure and function was explained in the 1970s by the biologist
Elizabeth Blackburn, who showed that telomeres generally become shorter as cells age.
She would share the 2009 Nobel Prize in Physiology or Medicine for the discovery.
Telomeres are regions of repetitive nucleotides that cap the ends of chromosomes,
protecting DNA from degradation as cells divide and replicate. First identified in
the 1930s, their structure and function was explained in the 1970s by the biologist
Elizabeth Blackburn, who showed that telomeres generally become shorter as cells age.
She would share the 2009 Nobel Prize in Physiology or Medicine for the discovery.
Better understanding of telomeres opened up exciting possibilities for researchers around the world, who worked to uncover their role in everything from cancer and genetic diseases to normal human aging. One was Belinda Needham, who in 2006 had just earned a PhD in sociology and was looking for a way to integrate recent advances in the biology of stress and aging into the study of population health.
“At the time there was evidence from human studies that people with shorter telomeres died sooner than people who, at the same age, had longer telomeres,” says Needham, who is now co-director of the Center for Social Epidemiology and Population Health and an associate professor of Epidemiology at the School of Public Health. “It made a lot of sense to people that it was like a clock that could help us understand how much longer people had to live.”
Needham thought telomeres might help explain disparities between health outcomes and life expectancy for people from different racial and socioeconomic backgrounds and reveal new ways to target interventions that could close those gaps. Working with a set of nearly 8,000 DNA samples collected as part of the CDC’s National Health and Nutrition Examination Survey, she confirmed the hypothesis that lower socioeconomic status correlated with shorter telomeres. But when she looked at the relationship between telomeres and race, she found that, despite having lower average life expectancies, African Americans actually tended to have longer telomeres than people of other races.
We can have greater confidence about a scientific claim if we see that many studies using a variety of techniques reached the same conclusion. But even then, we can never be sure that it won’t be disproven at some point in the future.
–Belinda Needham

“It was very surprising,” Needham says. “When you have what you think is a really great hypothesis that is going to be able to explain so much, and you get a finding that’s the complete opposite of what you expected, I think it’s very tempting to just say, ‘Well, that’s a fluke.’”
Needham would eventually come to see the unexpected result less as a setback than as a new avenue for inquiry. In the years since, she and other researchers have looked more closely at telomere length, including whether it actually does correlate with longevity in straightforward ways, as was once hoped. It was an early lesson in the surprising possibilities of the scientific method that has stayed with her.
“Many of the things that scientists study are really complicated and hard to observe,” Needham says. “We can have greater confidence about a scientific claim if we see that many studies using a variety of techniques reached the same conclusion. But even then, we can never be sure that it won’t be disproven at some point in the future.”
***
Throughout history, the process of discovery has always involved correcting mistakes, clarifying our understanding, and adding deeper shades of nuance.
For centuries, astronomers thought they had pinned down the position of the Earth relative to the sun and planets—until Nicolaus Copernicus came along and rearranged the model. It wasn’t until the 1990s that the scientific consensus solidified around anthropogenic global warming. Previously, some scientists had hypothesized that the planet’s average temperature was actually going down. Today, the emerging science of epigenetics is complicating our understanding of the genome’s role in heredity, health, and disease.
These changes in our understanding are features of science, not bugs.
Still, to laypeople and even to many scientists themselves, the shifting nature of scientific knowledge can be frustrating. That’s especially true in a field with real world implications like public health, which has been in the spotlight more than ever since the virus that causes COVID-19 began to spread. Throughout the pandemic, demand for information has often outpaced the ability of experts to provide answers, as new findings upended old assumptions and authorities repeatedly revised their guidance.
“When the first COVID-19 outbreak occurred, there was not much information about this virus,” says Chuanwu Xi, professor of Environmental Health Sciences and Global Public Health. “We used our previous experience dealing with viral outbreaks, like SARS in the 2000s and pandemic flu, and we believed that surface contamination was one of the major pathways of transmission.”

Eventually, Xi says, surveillance, testing, and lab work revealed that the SARS-CoV-2 virus, which causes COVID-19, was probably transmitted most readily in aerosolized respiratory droplets—and that changed the advice public health authorities gave the public.
“With airborne transmission, it’s really occurring more frequently in the indoor environment,” Xi says. “But even then, it depends on a lot of factors, including the viral load of an infectious person, temperature and humidity, the ventilation system, filtration, and frequency of the air exchange, which can dilute the viral particles in the air.”
![]() Public health experts now know that many of the safety practices we adopted early
on—deep-cleaning classrooms and subway cars, or disinfecting groceries and quarantining
the mail—are not necessary, while other measures, like masking and opening windows,
are much more effective than we originally realized. But those messages still haven’t
fully filtered into the public consciousness. And that points to a need for better
science communication, says Abram Wagner, a research assistant professor in the Department of Epidemiology.
Public health experts now know that many of the safety practices we adopted early
on—deep-cleaning classrooms and subway cars, or disinfecting groceries and quarantining
the mail—are not necessary, while other measures, like masking and opening windows,
are much more effective than we originally realized. But those messages still haven’t
fully filtered into the public consciousness. And that points to a need for better
science communication, says Abram Wagner, a research assistant professor in the Department of Epidemiology.
“We’ve all seen how messy communication was around, for example, wearing masks,” he says. “From a scientific perspective, we’re continually learning new information, so it makes sense that the message changes over time. But it becomes much more difficult to make people confident in the scientific process. That’s why I think being honest with people about the scope of what we know is important.”
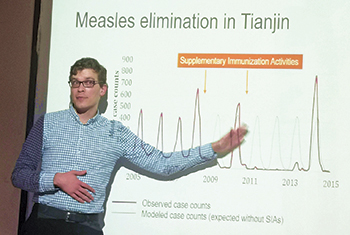
Earning public trust through transparency is especially crucial when it comes to vaccines, which can bring the pandemic to an end only if the public readily accepts them. Wagner, who studies vaccine hesitancy and public health messaging, points to measles as a cautionary tale. In 1998, a paper in the prestigious journal The Lancet linked the measles, mumps, and rubella vaccine to autism. The paper, it turned out, was based on fraud. But it took twelve years for the journal to retract it, and by that time the real-world damage was done. Measles had been declared eliminated in the US in 2000, but since then many parents, frightened by the possibility that the vaccine could trigger autism, have declined the shot, leading to flare-ups that peaked at nearly 1,300 cases in the US alone in 2019.
The long delay in retracting the paper points to a troubling lack of humility—and perhaps even a fear of being wrong—among scientific publishers, Wagner says. “I think there’s this idea that we need to project a certain amount of strength, and we need to position ourselves as experts, and that can be problematic.”
From a scientific perspective, we’re continually learning new information, so it makes sense that the message changes over time.
—Abram Wagner
That overconfidence—and the very human desire for clear answers—can be especially dangerous in the classroom, adds Needham, who argues that introducing more nuance into the science curriculum, even for elementary and high school students, could eventually help build trust in the scientific process among the general public.
“It can be appealing as an instructor to simplify the message,” Needham says. “But when you say, ‘You’re going to do this experiment, and you’re always going to get the same results,’ that can lead to the mistaken impression that that science is very neat and clean and that there is a predetermined correct answer.”
***
Lack of trust in science can be especially harmful for marginalized populations, who are sometimes subject to biases that were baked in to scientific theories by previous generations of researchers. It can be frustrating and confusing when science changes—but it can also be freeing, when it means shedding old prejudices.
“Science isn’t neutral, and it’s not objective,” notes Melissa Creary, assistant professor of Health Management and Policy. She works to instill that understanding in her students, who often arrive with a more idealistic view of science. “One of the things I ask them to do, when they’re thinking about articles they’re reading or about a research project they want to design, is to think also about the people who created the science.”
![]() With an aim toward anti-racism, Creary helps students think about how scientists’
backgrounds and points of view may have affected their methods and conclusions, to
question whether norms are rooted in facts or in beliefs, and to notice how findings
that seemed clear in a lab can look different in the real world. These issues, she
notes, are all well illustrated by her research, which focuses on sickle cell disease and the way in which this hereditary condition interacts with social reality to produce
different outcomes for people living with sickle cell disease.
With an aim toward anti-racism, Creary helps students think about how scientists’
backgrounds and points of view may have affected their methods and conclusions, to
question whether norms are rooted in facts or in beliefs, and to notice how findings
that seemed clear in a lab can look different in the real world. These issues, she
notes, are all well illustrated by her research, which focuses on sickle cell disease and the way in which this hereditary condition interacts with social reality to produce
different outcomes for people living with sickle cell disease.
Every high school biology student learns about sickle cell disease, a painful and sometimes fatal disorder of the red blood cells that’s most common in Black populations, because it’s a good example of Mendelian inheritance, in which a trait is controlled by a single gene with dominant and recessive forms. Sickle cell also shows how selective pressure can favor a mutation that would seem disadvantageous—in this case because, while two copies of the recessive gene cause the disease, individuals with a recessive copy on one chromosome and a dominant copy on the other are less susceptible to severe malaria than individuals with two dominant genes.
“It’s the standard monogenetic disease that everyone learns about,” says Creary, who is also a person living with sickle cell. “The first time I read about it was in my AP biology class in high school, and I was excited, because I was like, ‘Oh, this is me!’ I also knew that how it was described in the textbook, with laboratory jargon like ‘amino acids’ and ‘transcription’ and ‘translation,’ was different from how it worked in my body.”
That early intuitive understanding has informed Creary’s research in Brazil, which suggests that the ways in which the disease affects people vary according to the color of their skin, with darker people faring more poorly than those with lighter complexions.
Science isn’t neutral, and it’s not objective. One of the things I ask my students to do, when they’re thinking about articles they’re reading or about a research project they want to design, is to think also about the people who created the science.
—Melissa Creary

“Genetically, sickle cell looks the same for everyone,” Creary explains. “But the severity of the disease differs, because of what is happening in society. How you are perceived, how you are valued, actually helps or hinders health outcomes, even though a textbook would tell us that it’s the same for every person.”
Creary’s research contributes to a growing understanding of the ways gene expression is affected by the environment in which we develop, whether it’s the stresses of racial prejudice, chemical pollutants, or even the experiences of our parents and grandparents. Heredity, we are learning, is not as simple as AP biology textbooks once taught us.
***
In the 1990s, researchers working to map the human genome believed that as much as 98.5% of our nucleotides were non-functional. More recent research, however, has shown that while this so-called “junk DNA” doesn’t encode instructions for making proteins, it may determine how genes are expressed, effectively turning up the volume on some and lowering it on others.
“It’s a reminder that we need to be humble in science,” says Edward Ruiz-Narváez, an associate professor of Nutritional Sciences. “I always have been a little bit skeptical about genetic determinism—the idea that if we know all our genes, we will be able to predict everything. Now we’re learning so much about genetic regulation. Our concept of the genome has to change.”
Ruiz-Narváez is investigating how social and genetic factors may help explain the higher burden of disease in minority populations. He has recently begun thinking of the genome less as a set of genes than as “an emergent entity,” which isn’t fixed from conception, as scientists once believed.
It’s a reminder that we need to be humble in science.
—Edward Ruiz-Narváez

“There’s an instruction book that tells our genes when to turn on and how to react,” says Dana Dolinoy—NSF International Chair and professor of Environmental Health Sciences and professor of Nutritional Sciences—who studies the epigenome, the system of chemical tags that changes how DNA is transcribed.
 As a PhD student at Duke University in the mid-2000s, Dolinoy published a series of
influential papers based on work with obese rodents with yellow fur, known as “agouti
mice.” Their larger size and unusual coloring are caused by the activation of a gene
that is normally suppressed. Dolinoy and her colleagues discovered that the gene’s
expression in a baby mouse could be influenced by its mother’s diet and her exposure
to chemicals in the environment. The findings ran contrary to longstanding ideas about
heredity and evolution, which held that genes were passed from parent to child, altered
only by the occasional chance mutation.
As a PhD student at Duke University in the mid-2000s, Dolinoy published a series of
influential papers based on work with obese rodents with yellow fur, known as “agouti
mice.” Their larger size and unusual coloring are caused by the activation of a gene
that is normally suppressed. Dolinoy and her colleagues discovered that the gene’s
expression in a baby mouse could be influenced by its mother’s diet and her exposure
to chemicals in the environment. The findings ran contrary to longstanding ideas about
heredity and evolution, which held that genes were passed from parent to child, altered
only by the occasional chance mutation.
Today, Dolinoy realizes that her work represented a breakthrough, but it didn’t feel that way at the time. “It takes a very long time to see an experiment like that through,” she says, “and then—as it should be—your science goes into peer review and it is picked apart in every possible way.”

Dolinoy’s work helped create an entirely new field, environmental epigenetics, which investigates how environmental triggers, including diet, chemical exposures and stressful experiences, affect the epigenome. That has implications for preventing diseases, as well as for understanding and solving disparities in health outcomes between different populations. And, as researchers throughout the School of Public Health can attest, it’s always possible that this emerging science will overturn other concepts that everyone previously thought were solid.
“It can be hard to grasp that not only are there things that we don’t know, but also things that we do know that end up being wrong,” Dolinoy says. “The generation of knowledge is not a straight line. As technology changes and we’re able to ask different questions, good science can always evolve into something else.”
About the Author
Freelance writer Amy Crawford is a frequent contributor to Smithsonian and Michigan Alumnus. She lives in Ann Arbor.
- Interested in public health? Learn more today.
- Read New Vaccines for a Novel Virus: Insights from Coronavirus and Vaccine Experts in this issue of Findings.
- Support research and engaged learning at Michigan Public Health.